Acetic Acid
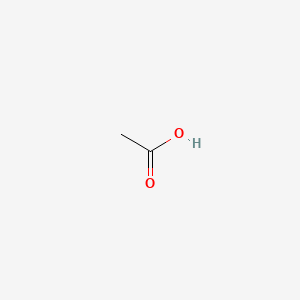
Formula: CH₃COOH — Vinegar (dilute), glacial acetic acid (pure), ethanoic acid
Appearance: Colorless liquid with pungent sour smell
Hazard: Flammable · Corrosive · Irritant
Properties
Colorless liquid with characteristic sharp, sour odor. Household vinegar is 4-8% acetic acid in water. A weak acid that partially dissociates in water. The conjugate base (acetate) is important in biochemistry. Miscible with water in all proportions. Freezes at 16.6°C - pure acetic acid is called “glacial” because it forms ice-like crystals in a cool room.
Historical Context
Vinegar is one of humanity’s oldest chemical products. The word comes from French vin aigre (“sour wine”). Babylonian records from 5000 BCE mention vinegar, and every wine-making civilization discovered it independently - bacteria convert alcohol to acetic acid when wine is exposed to air.
For millennia, vinegar was the strongest acid available to most people. Alchemists called it “philosopher’s vinegar” and used it to dissolve metals and minerals. Hannibal allegedly used heated vinegar to crack rocks blocking his army’s path across the Alps (though this may be legend).
The bacterium responsible, Acetobacter, was identified by Louis Pasteur in 1864. He showed that vinegar production was a biological process, not spontaneous, and developed methods to control it. Industrial production began in the early 20th century, and today acetic acid is a major industrial chemical.
The “volcano” experiment - baking soda and vinegar - has probably introduced more children to chemistry than any other demonstration. The reaction produces carbon dioxide gas: NaHCO₃ + CH₃COOH → CH₃COONa + H₂O + CO₂.
Obtaining
- Supermarket: White distilled vinegar (5% acetic acid) - cheapest option
- Cleaning vinegar: 6-10% - more concentrated, still safe
- Glacial acetic acid: Chemical suppliers only (99%+, corrosive)
Experiments
Volcano Reaction: The classic demonstration - combine with baking soda for vigorous CO₂ production. Add dish soap for more foam, food coloring for visual effect.
Egg in Vinegar: Dissolve an eggshell by soaking in vinegar for 24-48 hours. The calcium carbonate shell reacts with acid, leaving the flexible membrane intact. The “naked egg” demonstrates osmosis and chemical reactions.
Neutralization Demonstrations: React with bases to demonstrate acid-base chemistry. The reaction with milk of magnesia creates a beautiful color-changing rainbow when combined with pH indicator.
Making Sodium Acetate: Neutralize baking soda with vinegar, then boil off the water to produce sodium acetate for “hot ice” experiments.
Experiments using this chemical:
- Citric Acid Volcano - Alternative: vinegar works too
- Milk of Magnesia Rainbow - Neutralization demonstration
- CO₂ Density Demo - Generate CO₂ gas
- Hot Ice - Make sodium acetate from scratch
Safety
Moderate hazard — flammable; corrosive at high concentrations.
Incompatible with: Strong oxidisers (chromic acid, permanganates, nitric acid); strong bases; peroxides; reactive metals (aluminium, zinc — hydrogen gas generated)