Ammonia
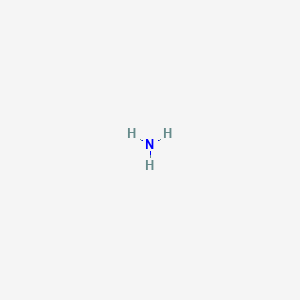
Formula: NH₃ (aq) — Ammonium hydroxide, household ammonia
Appearance: Clear liquid with a sharp, pungent odor
Hazard: Irritant · Pungent fumes · Weak base
Properties
Ammonia gas dissolved in water gives a mildly basic solution (5–10% solutions reach pH ~11–12). It is a weak base — unlike NaOH, it does not fully dissociate: NH₃ + H₂O ⇌ NH₄⁺ + OH⁻. This equilibrium makes it useful as a buffer component. A strong ligand: the lone pair on nitrogen coordinates readily to transition metal ions, often producing intensely colored complexes. Household ammonia (5–10%) is the standard laboratory grade for most experiments. Pungent fumes above open containers — always use in a well-ventilated area.
Historical Context
The name comes from Ammon, the Egyptian ram-headed deity whose temple in Libya was surrounded by camel caravans. Camel dung decomposing near the temple produced ammonium chloride crystals — sal ammoniacum, “salt of Ammon” — which, when heated, released the pungent gas we now call ammonia. Alchemists collected and used it for centuries without understanding its composition.
The chemistry was clarified in 1774 by Joseph Priestley, who isolated pure ammonia gas and showed it was a compound of nitrogen and hydrogen. But it was Fritz Haber’s breakthrough in 1909 — synthesizing ammonia directly from atmospheric nitrogen and hydrogen using an iron catalyst at high temperature and pressure — that changed history. The Haber-Bosch process enabled the industrial manufacture of nitrogen fertilisers, and is credited with feeding roughly half of today’s world population. It is arguably the most consequential chemical reaction of the 20th century.
Experiments
pH Test: A 5–10% ammonia solution reaches pH 11–12 with red cabbage indicator, giving a distinct blue-green/teal color — slightly different from sodium carbonate at a similar pH, because ammonia is a weaker base and its buffering properties create a slightly different ionic environment. Include it in The pH Landscape for comparison.
Copper Tetraamine Complex: Add ammonia solution drop by drop to a copper sulfate solution. First, a pale blue precipitate of Cu(OH)₂ forms; then, as more ammonia is added, it dissolves to give a spectacular deep royal blue/violet solution — the tetraamine copper(II) complex, [Cu(NH₃)₄(H₂O)₂]²⁺. This color is distinctly different from the sky blue of plain CuSO₄ and from all eight colors in The Many Colors of Copper. The color arises because NH₃ is a stronger-field ligand than H₂O, shifting the d-orbital energy gap and changing the absorbed wavelength.
Iron Hydroxide Precipitation: Add ammonia solution to ferric chloride — a red-brown Fe(OH)₃ precipitate forms, identical to using NaOH but gentler (the weak base prevents overshooting to very high pH). Similarly, adding to ferrous sulfate gives pale green Fe(OH)₂.
Ammonia Fountain (demonstration): Fill a dry flask with ammonia gas, invert it over a trough of water containing indicator. The gas dissolves so rapidly that a near-vacuum forms, drawing water up as a dramatic fountain. Demonstrates the extreme solubility of ammonia in water (~700 volumes of gas per volume of water at room temperature).
Experiments using this chemical:
- The pH Landscape — Basic test solution at pH ~11
- The Many Colors of Copper — Copper tetraamine complex [Cu(NH₃)₄]²⁺ deep royal blue (explore further)
Safety
Moderate hazard — irritant; sharp fumes at household concentrations.
Work outdoors or with good ventilation. Avoid breathing concentrated fumes — they irritate the respiratory tract. Do not mix with bleach (chloramines formed are toxic). Keep away from eyes; rinse immediately with water if splashed.
Incompatible with: Bleach/sodium hypochlorite (toxic chloramine gas — never mix); strong acids (vigorous neutralisation, ammonium salt formation and heat); mercury and silver compounds (explosive fulminates possible with concentrated ammonia); halogens; strong oxidisers