Ammonium Bicarbonate
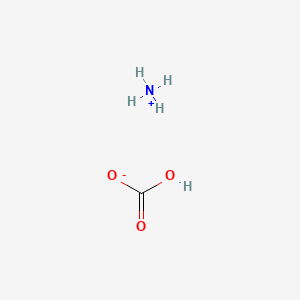
Formula: NH₄HCO₃ — Baker’s ammonia, hartshorn, ammonium hydrogen carbonate
Appearance: White crystalline powder with faint ammonia odor
Hazard: Irritant
Properties
White crystalline powder with a faint ammonia odor. Decomposes completely when heated into ammonia, carbon dioxide, and water - leaving no residue. Historically the primary leavening agent before baking powder. Best for thin, crisp cookies where ammonia can fully escape. Related to baking soda but produces more gas and leaves no alkaline residue.
Historical Context
Ammonium bicarbonate was the original chemical leavener, predating baking powder by centuries. The name “hartshorn” comes from its traditional source - the antlers of male deer (harts), which were heated in retorts to produce the compound. By the 18th century, it was being manufactured chemically.
Before hartshorn, bakers relied on yeast or beaten egg whites for leavening. Ammonium bicarbonate offered predictable, instant rise without the wait time of yeast fermentation. German and Scandinavian bakers particularly favored it for traditional cookies like springerle and speculoos.
The chemistry is elegant: NH₄HCO₃ → NH₃ + H₂O + CO₂. The compound decomposes completely into gases, leaving no residue (unlike baking soda, which leaves sodium carbonate). This creates exceptionally light, crisp textures - but the ammonia must fully escape, limiting its use to thin, dry baked goods.
When baking powder was developed in the 1840s, it largely replaced ammonium bicarbonate in most recipes. However, specialty bakers still prefer hartshorn for its unique textural effects in certain traditional cookies.
Preparation
Ammonium bicarbonate forms when ammonia and carbon dioxide meet in the presence of water — exactly what happens in your body during metabolism, and why it was originally collected near places where biological processes produced both gases. To make it at home, generate CO₂ by adding citric acid to baking soda in water, and bubble the gas through a cold solution of household ammonia: NH₃ + CO₂ + H₂O → NH₄HCO₃. Chill the ammonia solution in an ice bath during this step, since ammonium bicarbonate decomposes back into its gases above about 36°C. The white solid that precipitates or crystallizes upon cooling is ammonium bicarbonate, though yields from this method are modest.
Experiments
Complete Decomposition Demo: Heat gently in a test tube to show complete decomposition - the solid disappears entirely as it converts to gases. Demonstrates how a solid can become entirely gaseous. Use wet pH paper at the tube opening to detect ammonia.
Crispy Cookie Science: Bake cookies with ammonium bicarbonate vs. baking soda to compare textures. The complete decomposition creates a distinctively light, crisp texture. This shows how different leaveners affect food structure.
Gas Collection: Heat ammonium bicarbonate and collect the gases over water (CO₂ dissolves, leaving mostly ammonia) or use a two-stage collection to separate them. Demonstrates gas properties and solubility differences.
Experiments using this chemical:
- Food Chemistry - Leavening for crispy baked goods
Safety
Low hazard.
Incompatible with: Strong acids (rapid release of CO₂ and ammonia); strong bases (ammonia release); strong oxidisers