Ammonium Sulfate
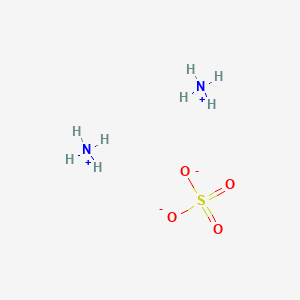
Formula: (NH₄)₂SO₄ — Diammonium sulfate, sulfate of ammonia, mascagnite
Appearance: White crystalline solid
Hazard: Irritant
Properties
White crystalline solid, highly soluble in water. One of the most important nitrogen fertilizers worldwide. Solutions are mildly acidic. Decomposes when heated, releasing ammonia. Used in fertilizers, food processing, and biochemistry for protein precipitation (“salting out”). Related to other ammonium salts; the sulfate is more stable than the nitrate.
Historical Context
Ammonium sulfate’s agricultural importance cannot be overstated. Before the Haber-Bosch process (1909) made synthetic ammonia available, nitrogen was agriculture’s limiting factor. Ammonium sulfate was one of the first widely-used nitrogen fertilizers, initially produced as a byproduct of coal gas manufacturing.
The compound became essential to biochemistry through “salting out” - the technique of precipitating proteins by adding high concentrations of ammonium sulfate. Discovered in the late 19th century, this remains a standard first step in protein purification. Different proteins precipitate at different ammonium sulfate concentrations, allowing crude separation.
The mineral mascagnite (natural ammonium sulfate) is rare, forming around volcanic vents where ammonia gases meet sulfuric acid. Most ammonium sulfate is now manufactured by reacting ammonia with sulfuric acid - a simple neutralization that produces one of the world’s most important chemicals.
Experiments
Protein Precipitation: Add to protein solutions to “salt out” proteins - high salt concentrations disrupt the hydration shell around proteins, causing precipitation. Used in biochemistry to purify proteins. Demonstrate with egg white solution.
Fertilizer Science: Use in plant growth experiments to demonstrate nitrogen requirements. Compare plant growth with and without ammonium sulfate supplementation. Shows how plants need nitrogen for protein synthesis.
Thermal Decomposition: Heating releases ammonia gas (detectable by smell or wet pH paper). Demonstrates thermal instability of ammonium salts: (NH₄)₂SO₄ → 2NH₃ + H₂SO₄.
Experiments using this chemical:
- Crystal Growing - Prismatic white crystals
Safety
Low hazard.
Incompatible with: Strong oxidisers; strong bases (ammonia released on heating); potassium chlorate and nitrate (hazardous when heated together)