Baking Soda
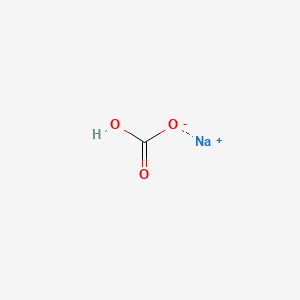
Formula: NaHCO₃ — Sodium bicarbonate
Appearance: White crystalline powder
Hazard: Not classified as hazardous
Properties
White crystalline powder, mildly alkaline (pH ~8.3). Occurs naturally as nahcolite. Decomposes at 50°C releasing CO₂ and water, leaving sodium carbonate. The workhorse of kitchen chemistry - reacts with any acid to produce bubbles. Used in baking as a leavening agent, in fire extinguishers, and as a gentle abrasive cleaner.
Historical Context
Sodium bicarbonate occurs naturally in mineral springs, and the ancient Egyptians used naturally occurring natron (a mixture of sodium carbonate and bicarbonate) for cleaning, mummification, and making glass. The pure compound was first prepared by Nicolas Leblanc in 1791 as part of his revolutionary process for making soda ash.
The baking revolution came in 1846 when Austin Church and John Dwight began manufacturing baking soda in New York. Their “Arm & Hammer” brand (named for the logo of Vulcan’s arm) became synonymous with the product. Before this, bakers relied on yeast or beaten egg whites for leavening - both slower and less reliable than the instant reaction of baking soda with acid.
The classic “volcano” experiment - baking soda plus vinegar - has introduced millions of children to chemistry. The dramatic fizzing reaction produces carbon dioxide gas while demonstrating acid-base neutralization. It’s perhaps the most replicated chemistry experiment in history.
Experiments
Volcano Reaction: The classic acid-base reaction - mix with vinegar (acetic acid) to produce CO₂ gas bubbles. Add dish soap for more foam. The reaction: NaHCO₃ + CH₃COOH → NaCH₃COO + H₂O + CO₂. Volcano demonstration
Fire Extinguisher: When heated, baking soda decomposes releasing CO₂: 2NaHCO₃ → Na₂CO₃ + H₂O + CO₂. This can smother small flames, demonstrating thermal decomposition and fire chemistry. Also shows how CO₂ is denser than air.
Experiments using this chemical:
- Citric Acid Volcano - Classic fizzing reaction
- CO₂ Density Demo - Invisible gas extinguishes candles
- Rainbow pH Indicator - Blue color at pH 8-9
- Food Chemistry - Leavening and fizzing reactions
Safety
Very low hazard — food-safe.
Incompatible with: Strong acids (vigorous CO₂ release — the expected reaction); aluminium metal in alkaline solution (hydrogen gas)