Borax
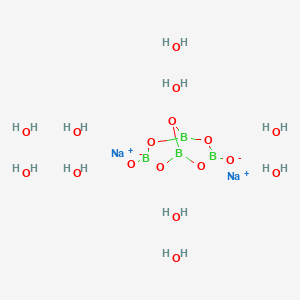
Formula: Na₂B₄O₇·10H₂O — Sodium tetraborate decahydrate, sodium borate, tincal
Appearance: White crystalline powder
Hazard: Harmful · Reproductive toxin · Irritant
Properties
White crystalline powder, mildly alkaline. Natural mineral (tincal) mined from dried lake beds. The borate ion is a gentle antiseptic and preservative. Used in detergents, cosmetics, and as a flux in metallurgy. Cross-links with polyvinyl alcohol to make slime. Related to boric acid - adding acid to borax solution produces boric acid.
Historical Context
Borax traveled the ancient Silk Road from Tibet to Europe, where it was prized by goldsmiths and glassmakers as a flux. The name derives from the Arabic bawraq or Persian burah. For centuries, the source of borax remained mysterious - Venetian glassmakers closely guarded their supply chains.
In 1776, the French chemist Jean-Pierre-Joseph d’Arcet discovered borax deposits in Tuscany, breaking the Venetian monopoly. The real transformation came in 1872 when Francis Marion Smith discovered massive borax deposits in Nevada and California’s Death Valley. His “Twenty Mule Team” wagons, hauling borax across the desert, became an American icon.
Borax’s ability to cross-link polymers was discovered in the 1960s, leading to the popular “slime” toy. The borate ion forms temporary bridges between polymer chains, creating a viscoelastic material that demonstrates non-Newtonian behavior.
Experiments
Slime: Dissolve borax in hot water; separately mix water with PVA glue. Combine both to create slime - the borate ions cross-link the polymer chains in the glue, creating a viscoelastic material that demonstrates polymer chemistry. Adjust ratios for different consistencies. Slime recipe
Crystal Ornaments: Make supersaturated borax solution and suspend pipe cleaner shapes overnight to grow spectacular crystals. The crystals form quickly and have a distinctive shape, perfect for making snowflakes or other decorative structures.
Experiments using this chemical:
- Crystal Growing - Fast prismatic crystals on pipe cleaners
- Slime - Cross-linked polymer with PVA glue
Safety
Moderate concern — reproductive toxin classification.
Incompatible with: Strong acids; aluminium and magnesium metal; basic lead acetate