Citric Acid
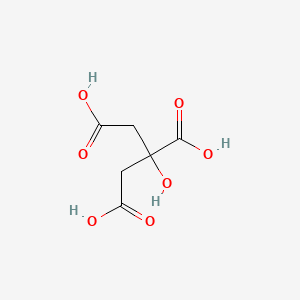
Formula: C₆H₈O₇ — 2-hydroxypropane-1,2,3-tricarboxylic acid, sour salt
Appearance: White crystalline powder
Hazard: Irritant
Properties
White crystalline powder with a sour taste. A triprotic acid (can donate three protons). Naturally abundant in citrus fruits. Excellent chelating agent for metals. Used as a preservative, flavoring, and cleaning agent. The citrate ion is an important intermediate in cellular metabolism (Krebs cycle). More acidic than acetic acid but weaker than mineral acids.
Historical Context
Citric acid was first isolated from lemon juice by the Swedish chemist Carl Wilhelm Scheele in 1784. For over a century, Italy dominated production, extracting the acid from citrus fruits - particularly lemons from Sicily. Italian producers carefully guarded their monopoly, and citric acid remained expensive.
The monopoly collapsed thanks to biochemistry. In 1917, American food chemist James Currie discovered that the mold Aspergillus niger could produce citric acid by fermenting sugar. By the 1920s, Pfizer had developed industrial fermentation processes that made Italian citrus extraction obsolete. Today, nearly all citric acid comes from fermentation, primarily in China.
Citric acid plays a central role in metabolism. Hans Krebs described the citric acid cycle (also called the Krebs cycle) in 1937, showing how cells extract energy from food. Every living cell runs this cycle, placing citric acid at the heart of biochemistry. Krebs received the Nobel Prize in 1953.
Preparation
Citric acid can be extracted from lemon juice using a simple precipitation method. Squeeze several lemons and filter the juice to remove pulp, then add calcium hydroxide (pickling lime) to the juice while stirring — calcium citrate precipitates as a white solid. Filter off the precipitate and wash it with water, then add dilute hydrochloric acid to dissolve the calcium and release the free citric acid: Ca₃(C₆H₅O₇)₂ + 6HCl → 2C₆H₈O₇ + 3CaCl₂. The calcium chloride stays dissolved, so evaporating the filtered solution yields impure citric acid crystals. The yield is modest but the process illustrates acid-base chemistry and selective precipitation beautifully.
Experiments
Fizzing Reactions: Combine with baking soda to create carbon dioxide gas, demonstrating acid-base reactions. Great for making “fizzy lemonade” or bath bombs. The reaction: citric acid + sodium bicarbonate → sodium citrate + water + CO₂.
Invisible Ink: Dilute citric acid solution can be used as invisible ink - write with it, let dry, then heat gently with a light bulb or iron to reveal brown writing. This demonstrates oxidation and caramelization of the acid. Video demonstration
Experiments using this chemical:
- Citric Acid Volcano - Fizzing with baking soda
- Invisible Ink - Heat-revealed writing
- Food Chemistry - Fizzing drinks and reactions
Safety
Low hazard — food-safe acid.
Incompatible with: Strong oxidisers (fire hazard in bulk); strong bases (neutralisation, heat); sodium bicarbonate and sodium carbonate (vigorous CO₂ evolution — often the intended reaction)