Corn Starch
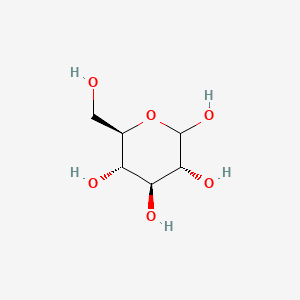
Formula: (C₆H₁₀O₅)ₙ — Cornstarch, cornflour (UK), maize starch
Appearance: Fine white powder
Hazard: Not classified as hazardous
Properties
A mixture of two glucose polymers: amylose (linear chains, ~25%) and amylopectin (heavily branched, ~75%). Amylose forms a helical structure whose interior cavity traps iodine (I₂) or triiodide (I₃⁻) molecules, producing a deep blue-black color — the starch-iodine test. Insoluble in cold water; gelatinizes when heated above ~65°C, forming a translucent gel. At high concentrations in cold water, forms a non-Newtonian dilatant fluid (“oobleck”) that becomes rigid under impact. Digested by amylase enzymes (saliva, pancreatic juice) to maltose and glucose.
Historical Context
Starch has been the primary human calorie source for most of recorded history — in grains, root vegetables, and legumes. Industrial extraction of corn starch was pioneered by Thomas Kingsford in 1842 in New Jersey; by the 1850s cornstarch had replaced arrowroot and wheat starch in most kitchens as a thickener and a laundry stiffener.
The iodine-starch reaction has been used as an analytical test since 1814, when Colin and Gaultier de Claubry independently discovered that iodine turns starch intensely blue-black. The reaction is exquisitely sensitive — starch concentrations too low to see by eye still give a visible color with iodine. This made it the indicator of choice for iodometric titrations throughout the 19th and 20th centuries, a role it still holds in the iodine clock experiment.
The non-Newtonian behavior of cornstarch suspensions (named “oobleck” after a Dr. Seuss story about a strange falling substance) became famous in physics demonstrations when it was shown that a person could run across a pool of the mixture. The phenomenon arises from dilatancy: under slow shear, starch granules rearrange to allow flow; under rapid impact, they jam together like a compressed powder, transmitting force rigidly.
Experiments
Starch-Iodine Indicator: Dissolve 1 teaspoon of cornstarch in 100 mL of hot water and let cool (the solution will be faintly cloudy). A few drops of this in the Iodine Clock experiment acts as the endpoint indicator — when free iodine appears at the moment ascorbic acid is depleted, the starch instantly turns deep blue-black. The reaction is reversible: boiling drives off the trapped iodine; adding sodium thiosulfate reduces I₂ to I⁻ and decolorizes the solution.
Oobleck: Mix approximately 2 cups of cornstarch into 1 cup of water. Stir slowly — it flows like a thick liquid. Strike it sharply with your palm or a spoon — it resists like a solid. The mixture returns to liquid when the force stops. This demonstrates shear-thickening (dilatant) non-Newtonian behavior.
Amylase Digestion: Confirm starch is present with the iodine test (dark blue). Then add a small amount of saliva (containing salivary amylase) and wait 5–10 minutes at body temperature. Re-test with iodine — the blue-black color is absent or much reduced, because the enzyme has hydrolyzed the starch to sugars.
Experiments using this chemical:
- Iodine Clock — Starch-iodide indicator for the sudden color-change endpoint
Safety
Very low hazard — food-grade.
Incompatible with: Iodine / iodide solution (intense blue-black color — the intended indicator reaction); strong oxidisers in bulk; moisture during storage (promotes mould growth — keep dry)