EDTA Disodium
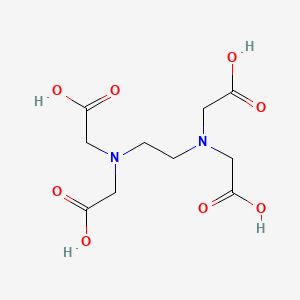
Formula: C₁₀H₁₄N₂O₈Na₂·2H₂O — Ethylenediaminetetraacetic acid disodium salt
Appearance: White crystalline powder
Hazard: Irritant
Properties
White crystalline powder, soluble in water. A hexadentate chelating agent - can bind metal ions at six points simultaneously, forming very stable complexes. Used in detergents, food preservation, and medicine (treating heavy metal poisoning). The stability constants vary by metal, allowing selective binding. Related to other chelators like citric acid but much stronger.
Historical Context
EDTA was first synthesized by Ferdinand Münz in Germany in 1935 while working for I.G. Farben. The compound was developed as a chelating agent to prevent metal-catalyzed degradation of industrial processes. Its ability to “sequester” metal ions made it valuable for water softening and preventing scale formation.
During World War II, EDTA found medical applications treating heavy metal poisoning. Its ability to form stable complexes with lead, mercury, and other toxic metals allows it to be safely excreted from the body. This “chelation therapy” remains a legitimate treatment for acute heavy metal poisoning.
The compound revolutionized analytical chemistry. Gerold Schwarzenbach’s development of EDTA titrations in the 1940s provided a simple, accurate method for measuring metal ion concentrations. Water hardness testing - determining calcium and magnesium levels - became routine rather than tedious.
Today, EDTA appears in countless products: detergents, food preservatives, cosmetics, and medical treatments. Its ability to bind metal ions makes it useful anywhere metal catalysis causes problems.
Experiments
Chelation Demo: EDTA binds strongly to metal ions - add to hard water to “sequester” calcium and magnesium, preventing soap scum. Demonstrates coordination chemistry and why chelating agents are used in detergents.
Metal Ion Titration: Use with indicator dyes like Eriochrome Black T to titrate water hardness. The EDTA binds metal ions, causing a color change that indicates the endpoint. This is a classic analytical chemistry technique.
Experiments using this chemical:
- Hard Water Demonstration - Titrate water hardness
Safety
Low-moderate hazard — irritant; potent metal chelator.
Incompatible with: Strong oxidisers; copper, iron, lead, and other heavy metals (rapidly chelated — the intended reaction); concentrated acids and bases; calcium compounds (calcium-EDTA complex formation)