Epsom Salt
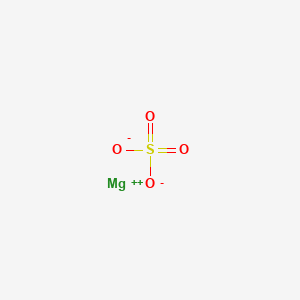
Formula: MgSO₄·7H₂O — Magnesium sulfate heptahydrate, epsomite
Appearance: Colorless crystalline solid
Hazard: Not classified as hazardous
Properties
Colorless crystalline solid, very soluble in water. Named after a spring in Epsom, England. Used medicinally as a laxative and in bath soaks. Forms distinctive needle-like monoclinic crystals unlike cubic salts. Related to other magnesium compounds; the magnesium can be precipitated as hydroxide with a strong base.
Historical Context
The story of Epsom salt begins in 1618 when a farmer named Henry Wicker noticed his cattle refused to drink from a spring on Epsom Common in Surrey, England. The bitter water, he discovered, had remarkable healing properties - it was a powerful laxative. Word spread quickly, and Epsom became England’s first spa town.
Nehemiah Grew isolated the salt in 1695 and named it sal Anglicum (English salt). It became a popular medicine throughout the 18th century, dispensed by apothecaries across Europe. The distinctive bitter taste (from magnesium) distinguished it from other “salts.”
The compound’s ability to form rapid, needle-like crystals made it a favorite for “crystal garden” experiments. When a saturated solution is painted on dark paper, the crystals grow visibly within minutes, their delicate structure branching like frost on a windowpane. This makes Epsom salt ideal for demonstrating crystallization to impatient young scientists.
Experiments
Crystal Needles: Grow delicate needle-like crystals that form quickly. Epsom salt crystals have a distinctive monoclinic structure different from cubic salt crystals. Can create “crystal gardens” by growing on porous materials.
Exothermic Dissolution: Unlike most salts, Epsom salt releases heat when dissolved (though less dramatically than calcium chloride). Measure temperature changes to demonstrate exothermic processes and compare with endothermic dissolution of ammonium chloride.
Experiments using this chemical:
- Crystal Growing - Quick needle-like crystals
- Natural Indicators - Beet juice color changes
Safety
Very low hazard — bath and food use.
Incompatible with: No significant incompatibilities at laboratory scale; calcium compounds form sparingly soluble magnesium calcium sulfate in some conditions