Hydrogen Peroxide
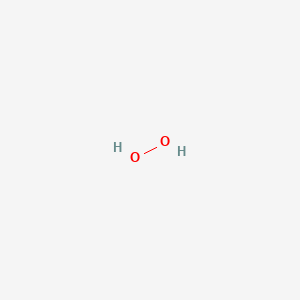
Formula: H₂O₂ — Hydrogen peroxide
Appearance: Clear, water-like liquid (3% pharmacy grade); can appear slightly viscous at higher concentrations
Hazard: Oxidiser · Irritant · Stronger concentrations corrosive
Properties
A pale blue liquid in pure form, appearing colorless at dilute concentrations. Decomposes to water and oxygen: 2H₂O₂ → 2H₂O + O₂. Decomposition is accelerated by heat, light, transition metal ions (especially Fe²⁺ — the Fenton reaction), manganese dioxide, and the enzyme catalase. Pharmacy-grade 3% solution is safe for general use. Pool-grade or “food-grade” 30–35% is a strong oxidizer requiring proper precautions. A powerful bleaching agent — discolors organic pigments and dyes by oxidation. Miscible with water in all proportions.
Historical Context
Hydrogen peroxide was first isolated by the French chemist Louis Jacques Thénard in 1818 by reacting barium peroxide with nitric acid — a reaction that no one had attempted before, because peroxides were barely understood. Thénard noted that the compound decomposed violently when catalysts were present and smelled faintly like ozone.
Industrial production came with the electrolytic process (1882) and then the modern anthraquinone process (developed in the 1930s–40s), which makes H₂O₂ indirectly via repeated oxidation and reduction of an anthraquinone carrier. Today, hydrogen peroxide is produced in millions of tonnes annually and is considered a “green” oxidant because it generates only water as byproduct.
The antiseptic use — bubbling on wounds to clean them — exploits catalase in blood and tissue, which rapidly decomposes H₂O₂ to produce the oxygen bubbles visible on a cut. Modern medicine has largely replaced it with better antiseptics, but it remains a household staple. In chemistry education, the catalytic decomposition demonstration — “elephant toothpaste” with concentrated H₂O₂ and a KI catalyst — has become one of the most spectacular and widely-watched chemistry demonstrations.
Experiments
Catalytic Decomposition (Enzyme): Add a small piece of raw potato or chicken liver to 3% hydrogen peroxide. Rapid bubbling of O₂ occurs — the enzyme catalase in living tissue decompose H₂O₂ as a protective mechanism. Boil the potato first and the reaction stops: heat denatures the enzyme, demonstrating the protein nature of enzymes.
Iron Oxidation Accelerator: Add a few drops of 3% H₂O₂ to a pale green ferrous sulfate solution and watch it turn yellow-brown as Fe²⁺ is rapidly oxidized to Fe³⁺. This demonstrates the same chemistry as rust formation but at observable speed. Connects to The Many Colors of Iron.
Bleaching Organic Dyes: Add a few drops of H₂O₂ to solutions of natural dyes (red cabbage juice, turmeric, tea) and observe the color fade as the oxidizer attacks the conjugated double-bond systems responsible for the color. Compare with dyes that resist bleaching (methylene blue is reduced, not oxidized, by H₂O₂).
Elephant Toothpaste (with higher concentration): Mix 30% H₂O₂ with potassium iodide solution (as catalyst) in a tall cylinder — a dramatic, rapid foam of oxygen bubbles erupts. The KI catalyzes decomposition; the dish soap traps the bubbles; the heat of the exothermic reaction warms the foam. Requires adult supervision and careful handling of 30% H₂O₂.
Experiments using this chemical:
- The Many Colors of Iron — Rapidly oxidise Fe²⁺ to Fe³⁺ to watch the color shift in real time
- Blue Bottle Reaction — H₂O₂ can substitute for shaking as the oxygen source to re-oxidize reduced methylene blue
Safety
Moderate hazard at 3%; high hazard at concentrations above 10%.
3% solution (pharmacy grade): Mild irritant. Safe for general use with normal care — wear gloves if prolonged skin contact is expected.
Above 10%: Corrosive; bleaches skin on contact. Wear gloves and eye protection. Avoid contamination with organic material, metal ions, or reducing agents, which can trigger vigorous decomposition.
Store in a cool, dark place in a loosely sealed container (decomposition produces O₂ which pressurises sealed bottles). Do not store in a sealed glass container — pressure build-up can cause rupture.
Incompatible with: Iron and iron salts (Fenton reaction — vigorous decomposition and reactive hydroxyl radicals); manganese dioxide, catalase, potassium iodide (rapid O₂ evolution); organic compounds at high concentration (fire and explosion risk); strong acids and bases (accelerate decomposition); copper, silver (catalytic decomposition)