Indigo Powder
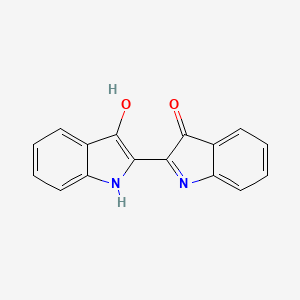
Formula: C₁₆H₁₀N₂O₂ — Indigotin, vat blue 1
Appearance: Deep blue powder
Hazard: Irritant
Properties
Deep blue powder, one of the oldest known dyes. Insoluble in water in its oxidized (blue) form. Must be chemically reduced to the soluble yellow leuco form for dyeing. The color develops as the dye oxidizes on the fabric when exposed to air. Used for denim jeans. Related to other vat dyes that work by similar redox chemistry.
Historical Context
Indigo is one of humanity’s oldest dyes, with evidence of its use dating back 6,000 years to Peru and 5,000 years to Egypt. The name comes from the Greek indikon, meaning “from India,” reflecting the importance of Indian indigo in the ancient dye trade. Roman naturalist Pliny the Elder wrote about Indian indigo in the 1st century CE.
The chemistry of indigo mystified dyers for millennia. The blue powder is insoluble in water - how could it dye fabric? Traditional methods, developed independently worldwide, used fermentation or chemical reduction to convert blue indigo into yellow leucoindigo, which is soluble. When fabric is removed from the vat, oxygen in the air re-oxidizes it back to insoluble blue, trapping the dye in the fibers.
In 1880, Adolf von Baeyer synthesized indigo artificially, though the process was too expensive for commercial use. BASF developed industrial synthesis by 1897, devastating the natural indigo trade in India but democratizing blue dye. Baeyer received the 1905 Nobel Prize partly for this work.
Today, indigo is synonymous with blue jeans. Levi Strauss’s original jeans used natural indigo; synthetic indigo now dyes billions of pairs of jeans annually.
Experiments
Redox Dyeing: Demonstrate oxidation-reduction chemistry by making “vat dyeing” solution. Reduce indigo with sodium dithionite in alkaline solution (it turns yellow-green), dip fabric, then watch it turn blue as oxygen re-oxidizes it when exposed to air. Indigo vat dyeing
Color Chemistry: Show how the same molecule can have different colors depending on its oxidation state. The reduced form (leucoindigo) is colorless/yellow while the oxidized form is the characteristic blue.
Experiments using this chemical:
- Indigo Vat Dyeing - Redox dyeing on fabric
Safety
Low hazard — intense staining dye.
Incompatible with: Strong oxidisers (bleach, hydrogen peroxide, permanganate — destroy the indigo chromophore, decolourising it; the basis of the leuco-indigo chemistry used in dyeing)