Methylene Blue
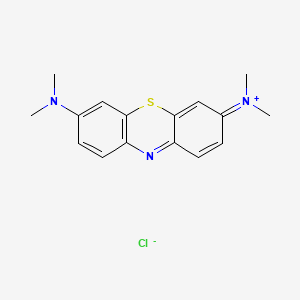
Formula: C₁₆H₁₈ClN₃S — Methylthioninium chloride
Appearance: Deep blue crystalline powder
Hazard: Harmful · Oxidising agent
Properties
Deep blue crystalline powder, very soluble in water. A thiazine dye that acts as a redox indicator - blue when oxidized, colorless when reduced. Used as a biological stain, antiseptic, and in the “blue bottle” demonstration. Can cross cell membranes and stain nucleic acids. Related to other thiazine dyes; historically used to treat malaria.
Historical Context
Methylene blue was the first fully synthetic drug used in medicine. Synthesized by Heinrich Caro at BASF in 1876, it was initially used as a textile dye. Its medical career began when Paul Ehrlich discovered its remarkable property of selectively staining biological tissues while leaving others colorless.
Ehrlich’s work with methylene blue led directly to his concept of the “magic bullet” - a drug that targets specific cells. He used it to stain and study the malaria parasite, then discovered it could actually treat malaria. This was revolutionary: a synthetic chemical could cure disease. Ehrlich’s methylene blue research laid the groundwork for modern chemotherapy and earned him the 1908 Nobel Prize in Medicine.
The “blue bottle” demonstration was developed as a chemistry teaching tool in the mid-20th century. Its reversible color change perfectly illustrates oxidation-reduction reactions, and generations of students have been captivated by the seemingly magical cycle of shaking (blue) and resting (colorless).
Experiments
Blue Bottle Experiment: Create a solution with glucose and sodium hydroxide, add methylene blue. Shake to turn it blue (oxygen oxidizes the dye), then watch it turn colorless as glucose reduces it. Repeat endlessly! Demonstrates redox chemistry beautifully. Blue bottle demo
Cell Staining: Use dilute methylene blue to stain cheek cells or onion cells for microscopy. The dye binds to DNA and RNA, making nuclei visible. Demonstrates selective staining and cell structure.
Oxygen Indicator: Add to a sealed container with a reducing agent - the blue color fades as oxygen is consumed. Can be used to demonstrate anaerobic conditions or test the effectiveness of oxygen absorbers.
Experiments using this chemical:
- Blue Bottle Reaction - Reversible redox demonstration
- Traffic Light Reaction - Multi-color indicator
Safety
Moderate hazard — potent stain; drug interactions.
Incompatible with: Strong reducing agents (reduced to colourless leuco form — the intended redox chemistry); strong oxidisers; iodides; persons taking MAO inhibitors or serotonergic drugs