Phenolphthalein
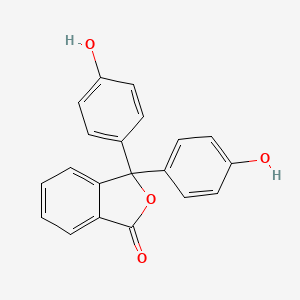
Formula: C₂₀H₁₄O₄ — Phenolphthalein
Appearance: White to pale yellow crystalline powder
Hazard: Irritant · Potential reproductive toxin
Properties
Sparingly soluble in water; readily dissolved in ethanol. Working solutions are typically 1% in ethanol. Colorless in acid and neutral solutions (pH < 8.2), turning vivid pink-magenta at pH 8.2–12, then colorless again above pH 12 due to a further structural rearrangement at very high base concentration. The sharp color transition at pH ~9 makes it ideal for strong acid/strong base titration endpoints. Commonly sold as a solution for direct use.
Historical Context
Phenolphthalein was synthesized in 1871 by Adolf von Baeyer — who later won the 1905 Nobel Prize in Chemistry for his work on organic dyes. Baeyer was systematically condensing aromatic compounds; reacting phenol with phthalic anhydride under concentrated sulfuric acid gave the intensely colored product.
For decades, phenolphthalein was sold as a laxative (most famously as Ex-Lax) because of its effects on intestinal smooth muscle. It was withdrawn from over-the-counter products in 1999 following animal studies suggesting possible carcinogenicity and reproductive toxicity at high doses, though the human relevance of those studies was disputed. As a laboratory indicator, the amounts used per experiment are negligible — a fraction of a milligram per titration.
The indicator remains the standard choice in school and university titrations because the pink endpoint is immediately visible, even in dilute solutions. The color transition is so sharp that a single drop of excess base can flip a 200 mL solution from colorless to vivid pink.
Experiments
Acid-Base Titration Endpoint: Add 2–3 drops of phenolphthalein solution to an acidic solution (vinegar, citric acid, or hydrochloric acid). Titrate drop by drop with sodium carbonate or sodium hydroxide. The endpoint is the first permanent pale pink — a single half-drop of excess base changes the color visibly. This is quantitative neutralization chemistry with a clear, unambiguous endpoint.
Comparison with Red Cabbage: Set up the same pH series used in The pH Landscape and add a few drops of phenolphthalein alongside red cabbage juice. Compare: red cabbage gives 8 distinct colors across the full range; phenolphthalein gives only colorless vs. pink, with a sharp switch at pH ~9. Both are useful — the choice depends on precision needed and pH range of interest.
Disappearing-Reappearing Ink: Write with phenolphthalein solution (colorless on paper). Spray with dilute sodium hydroxide — the writing turns vivid pink. Spray with dilute acid — it disappears again. Completely reversible and re-usable.
Experiments using this chemical:
- The pH Landscape — Comparison indicator alongside red cabbage juice
- Milk of Magnesia Rainbow — Alternative to universal indicator for neutralization
- Hard Water Titration — Endpoint indicator for EDTA titration
Safety
Moderate hazard — irritant; potential reproductive toxin at high doses.
Use dilute solutions (1% in ethanol). Avoid skin and eye contact with the ethanol solution. The amounts used in indicator experiments are negligibly small. Store away from heat and flame (ethanol solvent).
Incompatible with: Strong oxidisers (bleach, permanganate — oxidative decomposition of the dye); very strong base above pH 12 (turns colorless again — expected indicator behavior, not a hazard)