Potassium Alum
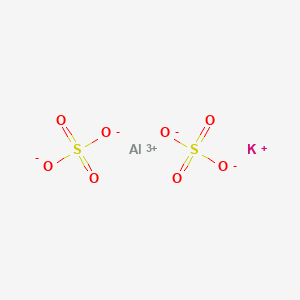
Formula: KAl(SO₄)₂·12H₂O — Potassium aluminum sulfate, potash alum, alum
Appearance: Colorless to white crystalline solid
Hazard: Irritant
Properties
Colorless to white crystalline solid, a double sulfate salt. Forms beautiful octahedral crystals that can grow very large. Historically used as a mordant in dyeing and in pickling. The aluminum ion makes solutions slightly acidic. Related to other alums (chrome alum, ammonium alum) which have similar crystal structures but different colors.
Historical Context
Alum has been prized since antiquity. The ancient Greeks and Romans used it as a mordant to fix dyes to cloth - without alum, most natural dyes would wash out. The textile industry’s demand for alum shaped medieval trade routes and politics.
The Papal States controlled Europe’s main alum source at Tolfa, Italy, discovered in 1462. Pope Pius II declared purchasing alum from non-Christian sources a sin, creating a lucrative monopoly that helped finance the Renaissance papacy. The English eventually broke free by developing alum works in Yorkshire in the 17th century.
Alum’s perfect octahedral crystals have made it the classic crystal-growing compound. In 1669, Nicolaus Steno studied alum crystals to develop his fundamental laws of crystallography - that crystal faces always meet at the same angles regardless of crystal size. These principles laid the foundation for modern crystallography and materials science.
Experiments
Giant Crystal Growing: Alum forms octahedral crystals that can grow to impressive sizes with patience. Use seed crystals and slow evaporation or cooling to grow single crystals several centimeters across. The crystals are transparent and beautifully geometric. Growing large alum crystals
Water Purification: Alum causes suspended particles to clump together (flocculation) and settle out of water. Demonstrate by adding to muddy water and watching it clear. This is the same chemistry used in water treatment plants.
Experiments using this chemical:
- Crystal Growing - Best beginner crystal, octahedral shape
- Natural Indicators - Mordanting for dye experiments
Safety
Low hazard — traditional household chemical.
Incompatible with: Strong bases (aluminium hydroxide precipitation, solution becomes strongly alkaline); alkali metals; strong oxidising agents