Red Cabbage Juice
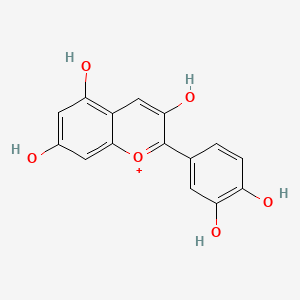
Formula: Mainly cyanidin-3,5-diglucoside and related anthocyanins — Red cabbage indicator, anthocyanin indicator
Appearance: Deep purple liquid when freshly extracted
Hazard: Not classified as hazardous
Properties
An aqueous extract of red cabbage (Brassica oleracea var. capitata rubra) containing a mixture of anthocyanins — primarily cyanidin glycosides conjugated to various sugars. Functions as a pH indicator spanning eight distinct color zones from pH 1 (crimson red) to pH 14 (yellow), one of the widest ranges of any single natural indicator. Not shelf-stable: use fresh or refrigerate for up to one week, freeze for longer storage. The color fades over time as anthocyanins oxidize.
Historical Context
Plant extracts that change color with acidity have been used since antiquity. Robert Boyle systematically documented the color changes of plant juices in acids and bases in the 1660s — this was among the first controlled observation of what we now call indicator behavior. Litmus, derived from certain lichens, became the standard laboratory indicator, but red cabbage remained a classic classroom demonstration because its striking purple juice, extractable by anyone with a kitchen, shows more color zones than litmus.
The compounds responsible, anthocyanins, are the pigments that color red cabbage, red wine, blueberries, black currants, and countless other plants. Their pH-dependent color change is a direct consequence of their molecular structure: the flavylium cation is positively charged and red in strong acid; losing successive protons as pH rises shifts the electron distribution through the conjugated ring system, moving the wavelength of maximum absorption progressively from green (transmitting red) through orange, yellow, and into the ultraviolet (transmitting yellow-green). Plants exploit this same pH sensitivity to regulate petal color — roses are red partly because their petal vacuoles are more acidic than those of blue hydrangeas.
Preparation
Standard method: Chop ¼ head of red cabbage (~200 g) and boil in 500 mL of water for 10 minutes. Strain the deep purple liquid. Yields approximately 400 mL of indicator. Use within a week refrigerated, or freeze in ice cube trays.
Quick method: Blend raw cabbage with cold water and strain immediately. Less anthocyanin extracted, but functional.
Indicator paper: Soak coffee filter strips in the indicator, let dry, and cut into strips. Portable pH test strips that develop color within 30–60 seconds.
pH Color Chart
| pH range | Color |
|---|---|
| 1–2 | Crimson / bright red |
| 3–4 | Pink-red |
| 5–6 | Violet-pink |
| 7 | Purple |
| 8–9 | Blue-violet |
| 10–11 | Teal / blue-green |
| 12–13 | Green |
| 14 | Yellow |
Experiments
The pH Landscape: The primary indicator for The pH Landscape experiment — test eight household acids and bases to build a complete visual pH scale. Each substance produces a distinct color from the chart above.
Milk of Magnesia Rainbow: Use in place of universal indicator in the Milk of Magnesia Rainbow experiment. The gradual neutralization of Mg(OH)₂ by vinegar shows a color gradient from blue-violet (basic) through purple (neutral) to pink-red (acidic).
Indicator Paper: Dry the indicator onto coffee filter strips for portable pH paper. Demonstrates how commercial pH paper is made.
Experiments using this chemical:
- The pH Landscape — Full acid-base color tour using anthocyanin indicator
- Milk of Magnesia Rainbow — Neutralization with color change
- Natural Indicators — Anthocyanin indicator chemistry and plant pigments
Safety
No hazard — food ingredient.
Incompatible with: Nothing hazardous. The purple color stains textiles and porous surfaces. Dilute bleach (sodium hypochlorite) will decolorize it.