Sodium Alginate
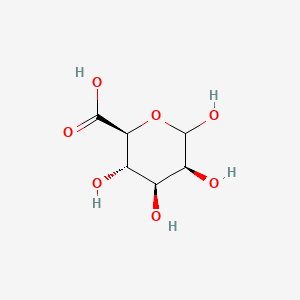
Formula: (C₆H₇O₆Na)ₙ — Algin, E401
Appearance: Brown to white powder
Hazard: Not classified as hazardous
Properties
Brown to white powder extracted from brown seaweed. Forms viscous solutions that gel instantly upon contact with calcium ions. The gel is heat-stable (won’t melt when heated). Used in food, textiles, and wound dressings. Works with calcium chloride or calcium lactate for spherification. Related to other hydrocolloids but unique in its calcium-reactivity.
Historical Context
The story of alginate begins with kelp harvesting, a practice dating back centuries along the coastlines of Ireland, Scotland, Japan, and China. Traditional uses included food thickening and medicinal applications. The chemistry was first investigated by British chemist E.C.C. Stanford in 1881, who named the substance “algin” from the Latin word for seaweed.
Commercial production began in the early 20th century, initially for textile printing. Alginate’s ability to form smooth, flexible films made it ideal for thickening print pastes. During World War II, alginate found new applications in wound dressings - its gelling properties create moist healing environments.
The molecular gastronomy revolution brought alginate to restaurant kitchens. In 2003, chef Ferran Adrià of elBulli popularized “spherification” - using sodium alginate and calcium to create liquid-filled spheres resembling caviar or ravioli with burst-in-your-mouth texture. This technique, essentially applying textile chemistry to food, became an icon of modernist cuisine.
Experiments
Spherification: Mix with water to create a solution, then drip into calcium chloride solution to form gel spheres - molecular gastronomy at home! The alginate cross-links with calcium to form instant gel membranes. Make “caviar” or edible water bottles. Spherification tutorial
Edible Films: Create thin, transparent films by spreading alginate solution and cross-linking with calcium spray. Demonstrates polymer chemistry and gel formation in a visually striking way.
Experiments using this chemical:
- Spherification - Gel spheres with calcium
- Food Chemistry: Spherification - Molecular gastronomy techniques
Safety
Very low hazard — food-grade thickener.
Incompatible with: Calcium, barium, and strontium salts (form gels or precipitates — the intended reaction in spherification); strong acids (precipitation at low pH); high salt concentrations (reduces gel strength)