Ferrous Sulfate
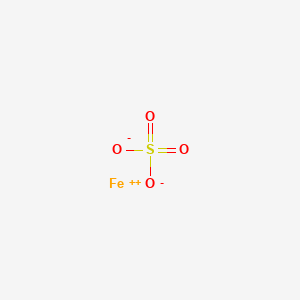
Formula: FeSO₄·7H₂O — Iron(II) sulfate heptahydrate, green vitriol, copperas, melanterite
Appearance: Pale blue-green crystalline solid
Hazard: Harmful · Irritant
Properties
Pale blue-green crystalline solid that oxidizes in air to yellowish ferric sulfate. Provides Fe²⁺ ions for ink making and reduction reactions. Used as an iron supplement, in water treatment, and historically for making iron gall ink. Related to ferric sulfate (Fe³⁺) - the color difference shows the oxidation state.
Historical Context
“Green vitriol” was known to the alchemists as one of the seven vitriols - the metal sulfates that formed colorful crystals resembling glass (vitrum in Latin). Its green color distinguished it from blue vitriol (copper sulfate) and white vitriol (zinc sulfate).
Ferrous sulfate’s greatest historical role was in ink making. Iron gall ink, made by combining ferrous sulfate with tannic acid from oak galls, was the standard writing ink in Europe from the 5th to 19th centuries. The Magna Carta, the Declaration of Independence, and countless manuscripts from Leonardo da Vinci to Johann Sebastian Bach were written with iron gall ink.
The chemistry is elegant: pale green Fe²⁺ ions combine with colorless tannic acid to form a barely visible solution. As the ink dries and the iron oxidizes to Fe³⁺, it forms an intense, permanent blue-black color. This “developing” process made forgery difficult and the ink nearly impossible to erase.
Experiments
Iron Gall Ink: Mix with tannic acid (from tea or oak galls) to create the historically important iron gall ink. The Fe²⁺ oxidizes to Fe³⁺ which forms an intense black complex with tannins. This was the standard writing ink for over a thousand years!
Oxidation States: Observe the green Fe²⁺ solution turn yellow-brown as it oxidizes to Fe³⁺. Add hydrogen peroxide to speed oxidation. Add vitamin C (ascorbic acid) to reduce it back to green. Demonstrates redox chemistry with visible color changes.
Prussian Blue Synthesis: React with potassium ferricyanide to instantly produce the intense Prussian blue pigment. This demonstrates how the same element in different oxidation states can create striking colors.
Experiments using this chemical:
- The Many Colors of Iron - Fe²⁺ pale green, rust formation, iron gall ink
- Crystal Growing - Blue-green crystals (oxidizes in air)
- Prussian Blue Synthesis - Intense blue pigment
- Iron Gall Ink - Classic iron-tannin ink
- Natural Indicators - Dark color with anthocyanins
Safety
Moderate hazard — harmful; keep out of reach of children.
Incompatible with: Strong oxidisers (Fe²⁺ oxidised to Fe³⁺ — changes colour from green to yellow-brown); strong bases (iron hydroxide precipitation); hydrogen peroxide (Fenton reaction generating reactive hydroxyl radicals); permanganate